A new study, just published in Nature Communications, led by researchers in the Roselló-Díez Lab has found a self-repair system inside the developing skeleton that helps bones recover from early growth disorders.
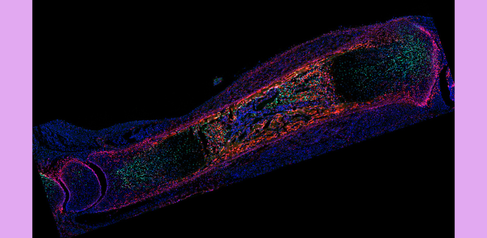
The team discovered that when cartilage growth is interrupted, a small population of stromal cells surrounding the cartilage can switch roles. Stromal cells are a type of connective tissue cell that usually provide structure and support, but here they act more like stem cells, transforming into cartilage-producing cells that restore normal bone growth.
These “repair cells,” marked by a gene called Gli1, then remain in the body as long-lived progenitors that continue supporting bone growth throughout childhood.
“The system is remarkably plastic, it’s like the skeleton has a built-in backup plan,” says senior author Professor Alberto Roselló-Díez. “When growth goes off track, these cells step in to protect development.”
By gently blocking and manipulating cartilage growth in developing mice, they were able to see how the tissue responded to stress. These findings revealed how these fetal cells not only creates the cartilage that forms in juvenile animals, but are also part of the skeleton’s natural resilience system, able to repair cartilage and restore normal growth.
This understanding of how the fetal skeleton repairs itself could transform how childhood growth disorders are treated, as well as growth-plate injuries, and even cartilage loss in adults. The growth suppressor that the team used to slow down cartilage growth is abnormally abundant in the cartilage of people with achondroplasia (short-limb dwarfism), suggesting that the discovered repair mechanism could be used to treat this condition.
“If we can learn how to reactivate these pathways in a non-invasive way, we might one day repair cartilage without transplants,” says Roselló-Díez.
The study, Gli1-expressing stromal cells are highly reparative precursors of long-lived chondroprogenitors in the fetal murine limb, has just been published in Nature Communications.
Taken from an ARMI article Discovery reveals the skeleton’s hidden repair system
